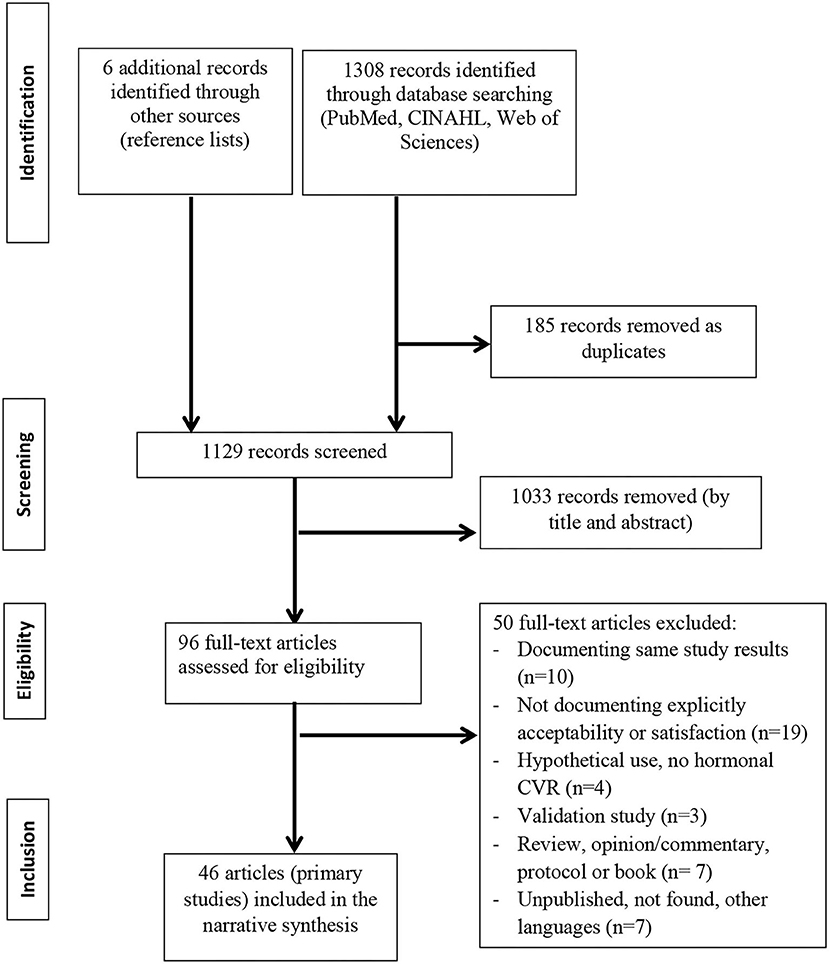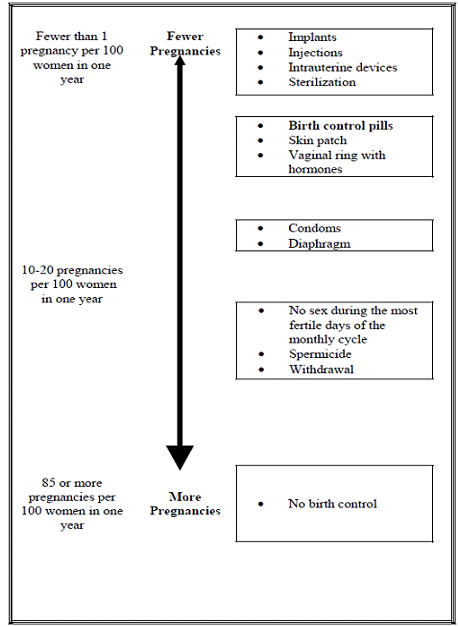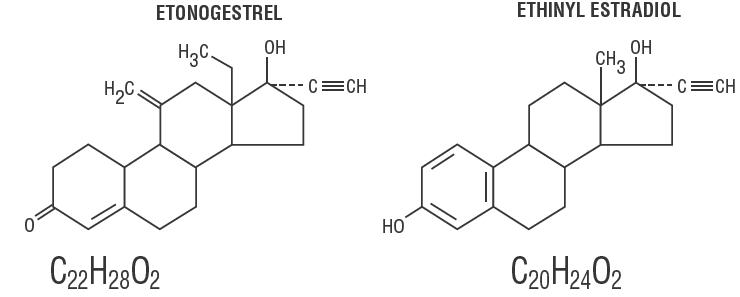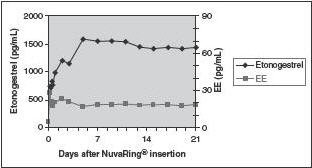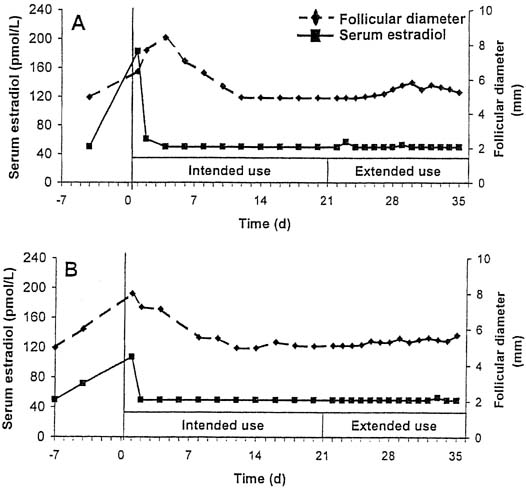
FDA Approves EluRyng (etonogestrel/ethinyl estradiol vaginal ring), the First Generic NuvaRing-CliniExpert

These highlights do not include all the information needed to use ELURYNG safely and effectively. See full prescribing information for ELURYNG. ELURYNG ™ (etonogestrel and ethinyl estradiol vaginal ring) Initial U.S. Approval: