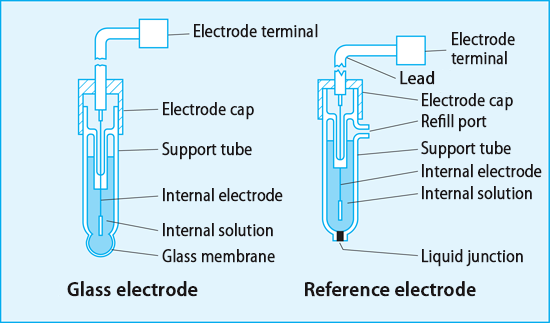
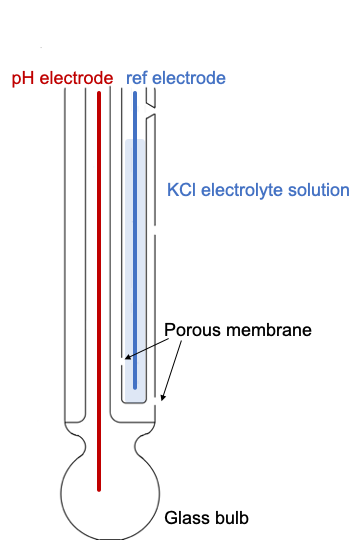
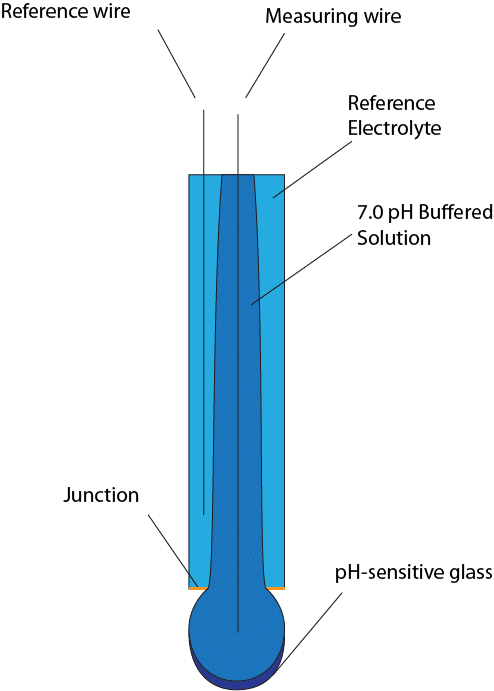
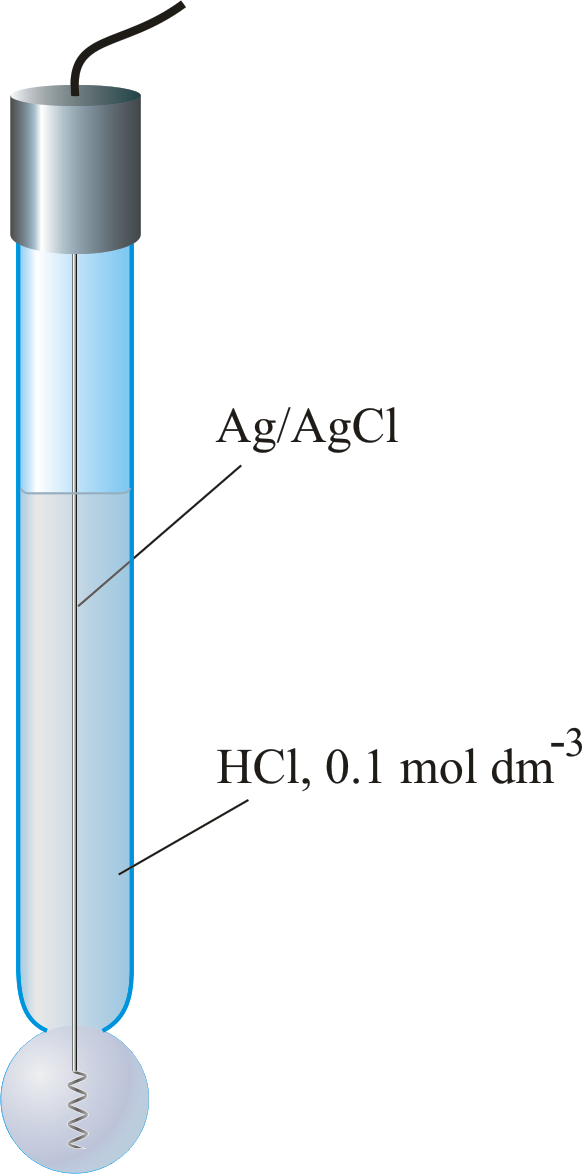
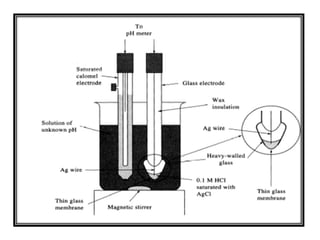
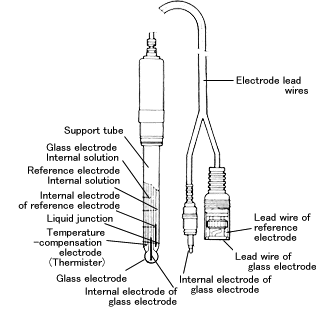
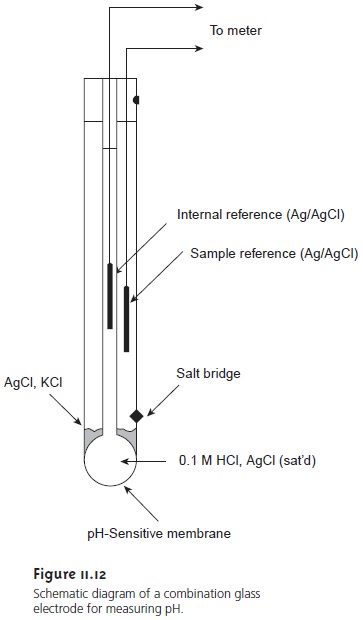
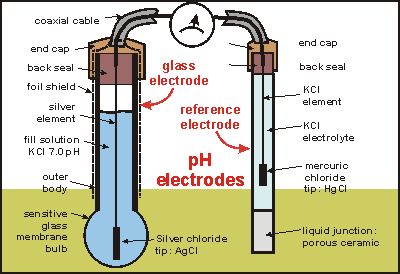
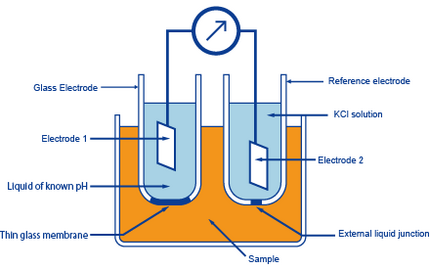
a. Show with the aid of a form of the Nernst equation that when using a pH sensitive glass electrode and reference electrode that the measured potential is proportional to pH. b.

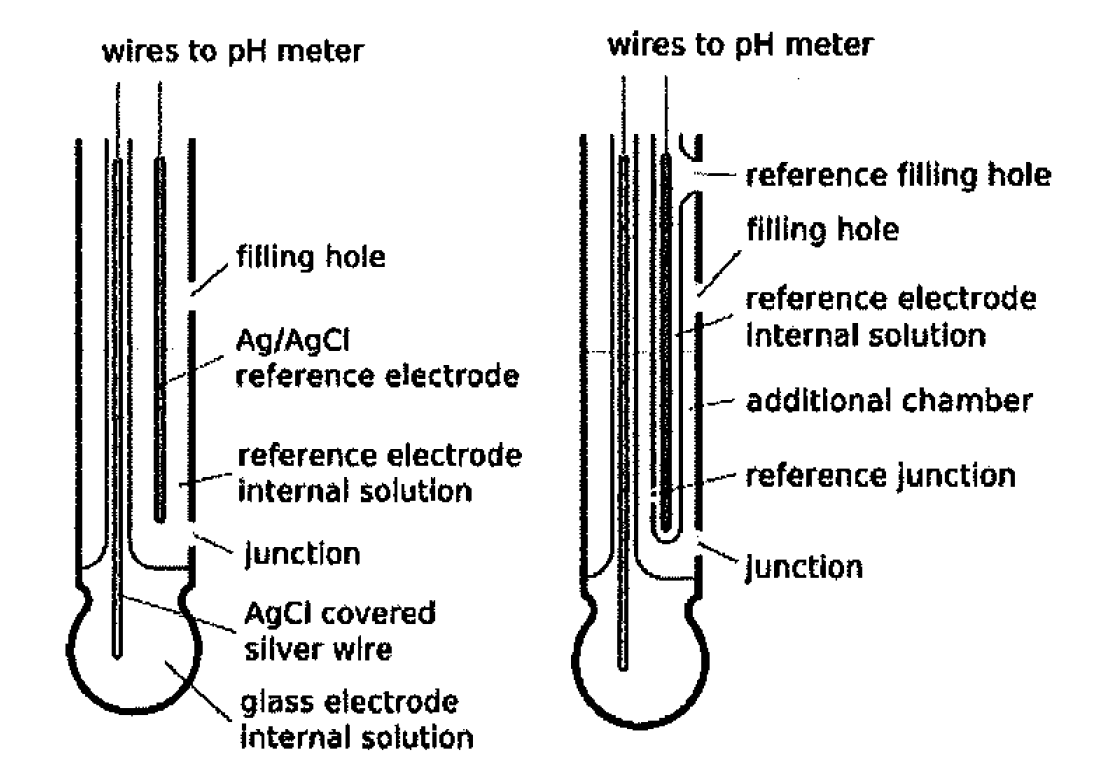
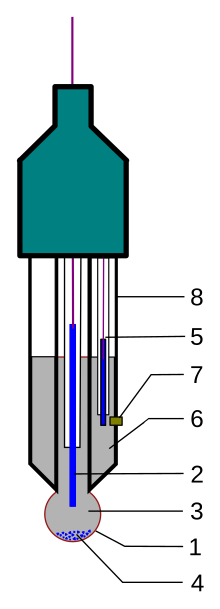
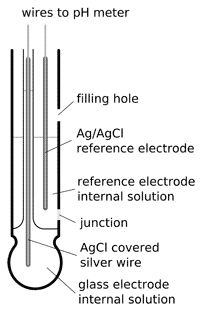
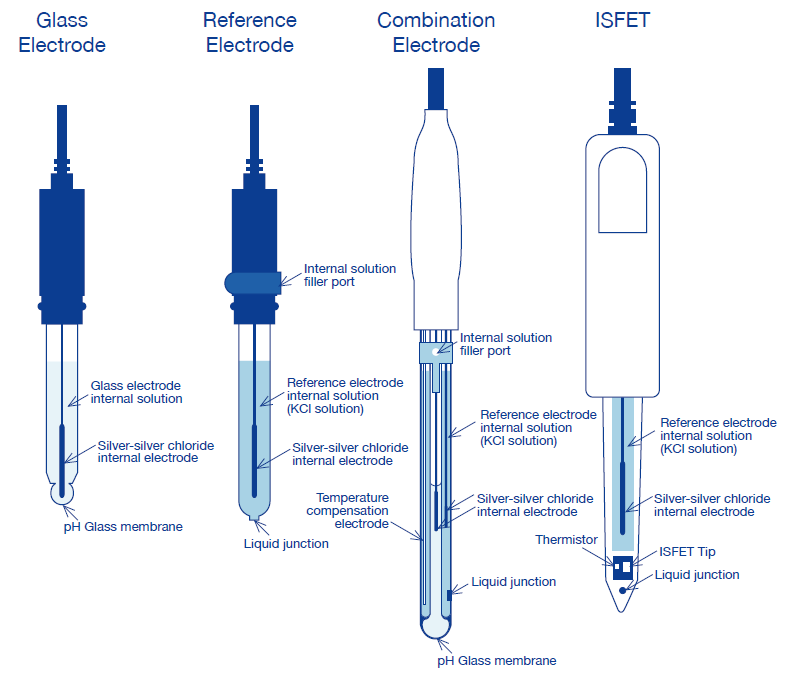
Draw schematically a combined pH glass electrode and explain the working principle. | Homework.Study.com